An often overlooked cause of genetic disease
by Elisabeth Paymal
Often overlooked because they might not alter the amino acid production, splicing defects can play an important role in rare genetic pituitary gland diseases.
Several clinical endocrinologists and bioinformaticians, led by Sally Camper and Jacob Kitzman, Professors of Human Genetics at the University of Michigan Medical School (UMMS), came together to further investigate splicing defects and their roles in rare genetic hormonal diseases. Cathy Smith, a PhD student, established a catalog of predicted variants that could either be tolerated or disease causing.
The pituitary gland is a very important center for hormonal activity. It produces several different hormones which control growth and the functions of other endocrine glands. Specific to the pituitary gland is POU1F1 (also known as PIT1), a transcription factor critical for the development of three of the hormone-producing cell types: growth hormone, prolactin, and thyroid stimulating hormone. Most of the time, a mutation in POU1F1 is recessive, but there are some cases when the variants have a dominant effect. Either genetic cause usually results in multiple hormone deficiencies.
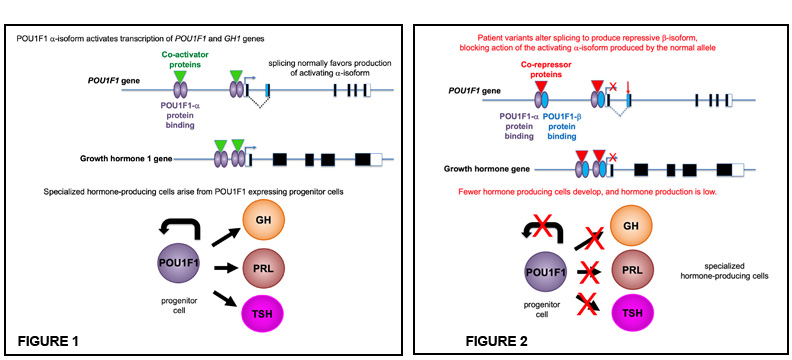
Figures: POU1F1 acts as a dimer and binds to its own gene and the growth hormone to activate transcription (figure 1). The patient variants shift splicing to producing the repressor isoform, which can block the action of the alpha isoform produced from the normal gene copy of POU1F1 (figure 2). This results in less POU1F1 transcription, fewer hormone producing cells, and less growth hormone.
In a recent study, Camper and colleagues discovered splicing defects in POU1F1 in six families with children who presented with congenital hormone deficiency. Splicing is the process by which the introns (the noncoding regions of genes) are removed from the primary messenger RNA transcript, and the exons (the coding regions) are joined together to form the mature mRNA that will, in turn, make the protein. In the pituitary gland, two forms of POU1F1 are produced by alternate splicing: the predominant one is a transcriptional activator (POU1F1-alpha) and the other one, that is usually a rare form, is a repressor (POU1F1-beta). When splice enhancer or repressor sites are mutated, the POU1F1 gene mostly produces the transcriptional repressor which shuts down hormone production, causing extreme short stature and hypothyroidism.
Camper’s research piqued the interest of Jacob Kitzman, Assistant Professor of Human Genetics and of Computational Medicine and Bioinformatics at the UMMS. Kitzman suggested making and testing every possible mutation, to find others that could disrupt splicing similarly to the patient variants, and to learn what factors the splice-disruptive variants had in common. Kitzman and his graduate student Cathy Smith applied experimental mutagenesis techniques and bioinformatic analyses that Kitzman had previously developed for cancer variant analysis. “Splicing effects are often difficult to predict, so we thought this would be a great opportunity for some of the approaches my group has developed to systematically test thousands of variants at a time,” said Kitzman.
The team found several variants that could disrupt splicing while not affecting the altering the amino acid sequence of the protein. The scientists established a catalog of gene variants that affects how the transcription factor is spliced in the pituitary gland. This catalog establishes whether a particular variant is predictive of a disease. “We all carry many variants in our genes, and it is difficult to identify those that are deleterious from those that are benign. For precision medicine, we need to understand the possible effect of each variant, and this catalog is very useful for this,” explained Camper.
This team then asked collaborators for a list of variants from their patients’ sequences. They found two families with variants in POU1F1 that matched the catalog of experimentally determined splice disruptive changes that do not alter the amino acid sequence. This strongly confirms that the Camper and collaborators’ catalog reliably identifies variant splicing that causes diseases.
This study also allowed comparison of several software packages that predict splicing outcomes, and one stood out as the best predictor. “This validation is more broadly interesting for people who work on any disease. It gives them more confidence in the predictions from state-of-the-art software, and in the quality of their data and results,” said Camper.
One of the challenges with studying rare diseases is to find patients, requiring extensive collaborations. For example, pituitary hormone deficiency occurs in approximatively 1 in 4,000 live births. Approximately 3% of the cases are due to mutations in the alpha isoform of POU1F1. Camper’s collaborative study relies on six unrelated families, three from France, one from Brazil, one from Germany, and one from Argentina. The collaboration involved three former post-doctoral fellows from Camper’s lab who, upon returning to Marseille, France, São Paulo, Brazil, and Buenos Aires, Argentina, joined large pediatric hospitals. The French group belonged to an endocrinologists’ network in francophone countries, including in north Africa. They were able to identify three families with variants that affected POU1F1 splicing for this study.
“When working on uncommon disease, it is very helpful to establish strong collaborations to identify and enroll patients whose participation is essential. The results from our study are much more convincing thanks to the six unrelated families who presented the same type of genetic defect,” said Camper. “Nobody could do that by themselves.”
In another recent collaborative study that screened 170 Argentinean patients for mutations in known hypopituitarism genes, only 15% of the patients had a mutation in a known gene. Camper’s team is now planning to sequence all the DNA of these patients and hopes to discover additional genes that might play a role in these diseases.
Camper and Kitzman are faculty members of the U-M Center for RNA Biomedicine.
Cited publications:
Gergics et al., High-throughput splicing assays identify missense and silent splice-disruptive POU1F1 variants underlying pituitary hormone deficiency, The American Journal of Human Genetics (2021), https://doi.org/10.1016/j.ajhg.2021.06.013.
Vishnopolska et al., Comprehensive identification of pathogenic gene variants in patients with neuroendocrine disorders, Journal of Clinical Endocrinology and Metabolism (2021), doi: 10.1210/clinem/dgab177.