Nils Walter and Adrien Chauvier publish new review article – and it’s about time… literally!


Center for RNA Biomedicine Co-Director Nils Walter, Ph.D. is excited about sharing some news about yet another fascinating role RNA plays in gene regulation – and it’s about time… literally.
In a new review article, “Regulation of bacterial gene expression by non-coding RNA: It is all about time!” published January 10, 2024 in the journal Cell Chemical Biology, Walter and his colleague Adrien Chauvier, Ph.D., at the Single Molecule Analysis Group and Center for RNA Biomedicine, Department of Chemistry, University of Michigan, discuss the vital role that speed plays in the process of non-coding RNA (ncRNA) folding in bacteria.
Bacteria are curious little things. Not to mention one of nature’s most highly-skilled quick-change artists. It’s a game of survival of the fittest. Bacteria are born assimilaters – adapting readily to their surrounding environment, and this requires the ability to change how their genes operate.
In a living cell, RNA is made from DNA through a process called transcription – the first step in gene expression. This RNA can then be turned into proteins, and the entire process is overseen by a watch-dog of an enzyme, RNA polymerase( RNAP).
Of all RNAs sythesized, there are two types: ones that helps make proteins (coding RNAs); and ones that don’t (non-coding RNAs, or ncRNAs). However, the non-coding RNAs are crucially important as they help regulate the genes, and control various and sundry other processes like cell division, the degree to which bacteria react to environmental changes, and so on.
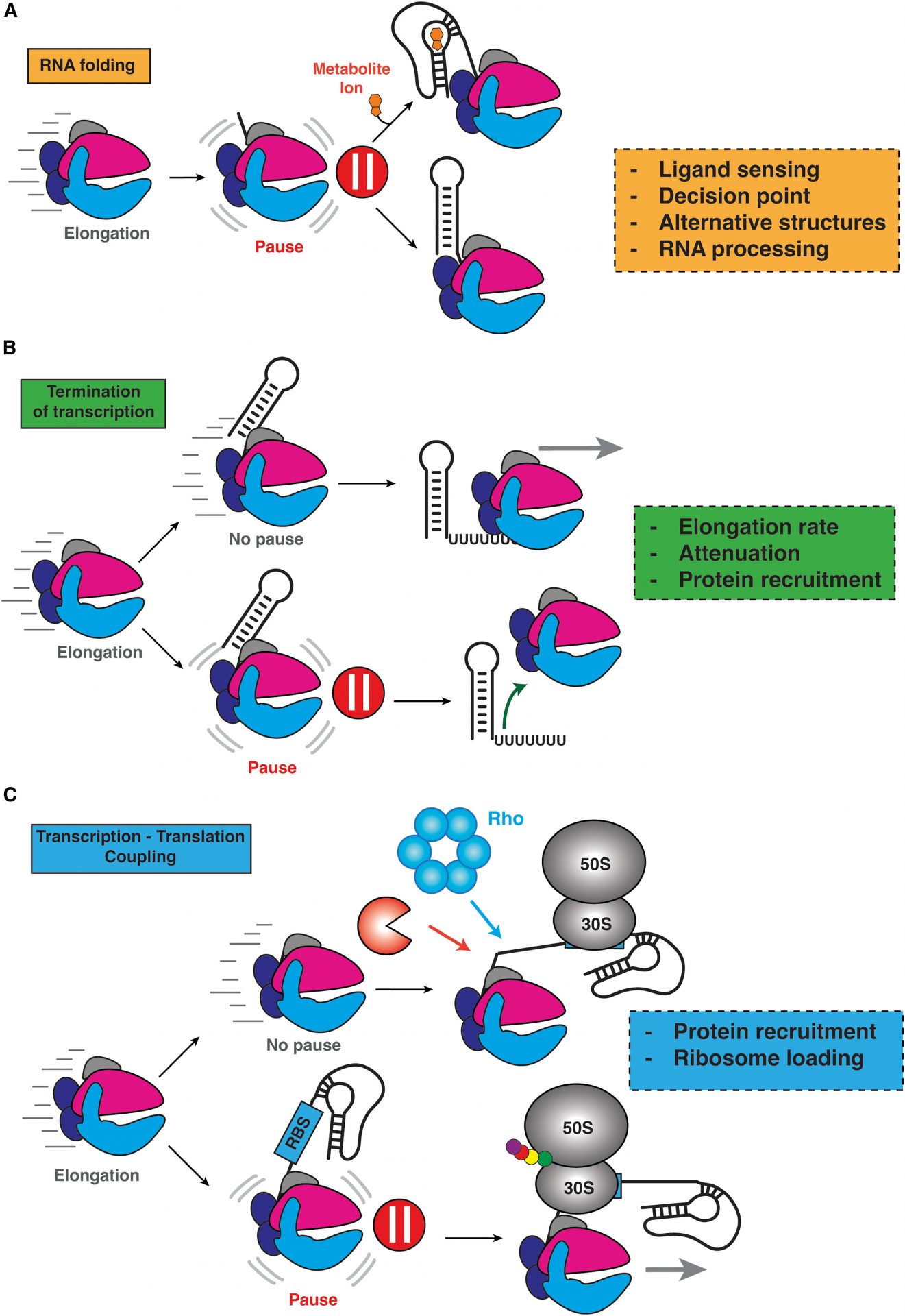
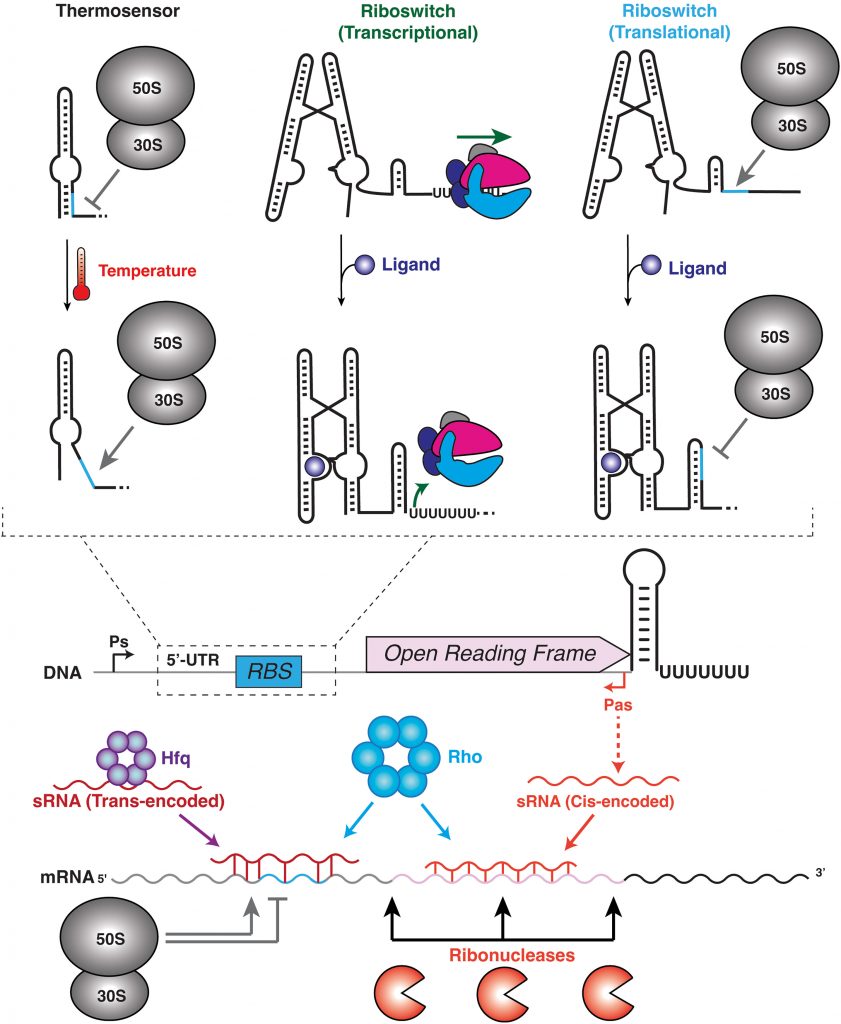
In this examination, Walter and Chauvier explore how non-coding RNAs can fold into different structures when they are created, and that this process can markedly affect exactly how they function. The speed of this process matters greatly since changes can lead to serious problems for the bacteria. It’s a delicate balance of processes – mainly timing – that could mean life or death for these single-cell stalwarts.
They further emphasize just how incredibly complex ncRNAs are, and that uncovering how they fold while being made and how this is directly related to their function will help us better understand how bacteria survive and evolve – knowledge that could vastly improve how we design and use antibiotics to treat bacterial infections.
Walter and Chauvier revel in this exciting field of study that holds terrific promise for the future for finding potential links to diseases and also be valuable for developing new treatments. Indeed, a booming new destination along the ever-growing RNA therapeutics superhighway that’s definitely worth a visit.
Science. Accelerating.
To read the full review article, visit: https://doi.org/10.1016/j.chembiol.2023.12.011