- This event has passed.
RNA Innovation Seminar: Andrey S. Krasilnikov, PhD, Penn State University
Monday, September 21, 2020 @ 4:00 pm - 5:00 pm

“Structures of eukaryotic RNases MRP/P RNPs reveal RNA-driven protein remodeling”
Andrey Krasilnikov, Associate Professor of Biochemistry and Molecular Biology, Center for RNA Biology, Pennsylvania State University
Keywords: Ribozymes, RNase P, RNase MRP, ribonucleoprotein complexes, RNA-driven protein remodelling
REGISTRATION REQUIRED:
https://umich.zoom.us/webinar/register/WN_obckKUCLT4mXI7kPskzc-Q
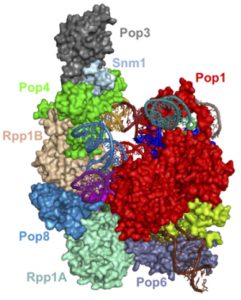
Abstract: Ribonuclease (RNase) P is a ribozyme-based catalytic ribonucleoprotein complex involved primarily in the maturation of tRNA in all three domains of life. In the course of evolution, the size and complexity of RNase P grew as the catalytic RNA moiety recruited additional protein components. In eukaryotes, the RNase P lineage has split, giving rise to a related RNP enzyme called RNase MRP, which shares multiple structural features (including most of the protein components) with the eukaryotic RNase P, but has a distinct and non-overlapping specificity.
We report the recently solved cryo-EM structure of the 450 kDa yeast RNase MRP holoenzyme and compare it with the structure of its progenitor RNP, RNase P. We show that, surprisingly, several of the proteins shared by RNase MRP and RNase P undergo RNA-driven structural remodeling, allowing the same proteins to function in distinct structural contexts. This remodeling, combined with altered peripheral RNA elements, results in the functional diversification of the two closely related RNPs, in spite of the structural conservation of the nearly identical catalytic cores, demonstrating structural underpinnings of the acquisition of new functions by catalytic RNPs.

